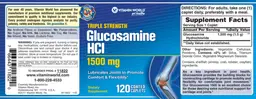
MSM with Glucosamine Sulfate
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 50% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Recommendations: Take 1 to 3 UltraCaps daily.
⚠️ Warnings & Precautions
If pregnant, nursing, or taking prescription drugs, consult your healthcare practitioner prior to use.
🧪 Formulation Notes
Contains no sugar, yeast, wheat, gluten, corn, soy, dairy products, artificial coloring, artificial flavoring, or preservatives.
Additional Information
MSM is a bioavailable form of sulfur, which is necessary for the proper function of the body's skin, hair and nails, connective tissue, amino acid production, and immune system. Sulfur must be consumed each day for healthy function and growth of the body. Glucosamine sulfate is up to 98% absorbable, which makes it more readily available to the body to support healthy joint cartilage. Clinical studies show it's a key building block for connective tissue and cartilage.
LI77559.B03
PREMIER SCIENCE
Supports joints and joint cartilage
This product contains natural ingredients; color variations are normal.
Product Details
Research Evidence
Compare Similar Products