Glucosamine Sulfate 500 mg
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 100% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
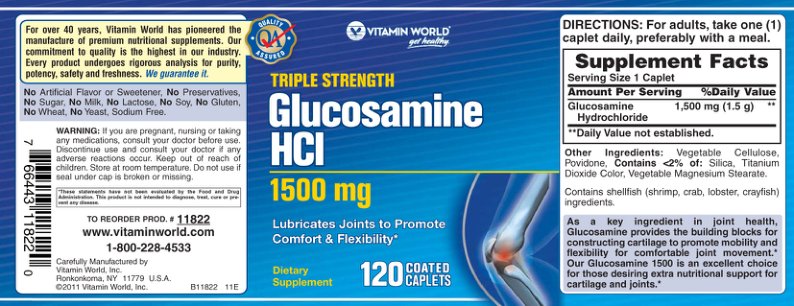
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
INDICATION: Helps relieve osteoarthritic symptoms and joint pain. Helps to build healthy cartilage and protect against deterioration. DIRECTIONS: Adults: Take 1 caplet 3 times daily, or as directed by a physician or health care practitioner. Use for a minimum of 3 months to see beneficial effects.
⚠️ Warnings & Precautions
KEEP OUT OF REACH OF CHILDREN.
Warning: Consult a health care practitioner prior to use if you have diabetes. Consult a health care practitioner if symptoms persist.
Contraindications: Do not use if you have shellfish allergies.
Do not use if pregnant or breastfeeding.
DO NOT USE IF SEAL UNDER CAP IS BROKEN.
🧪 Formulation Notes
Each 1 caplet contains 71 mg of potassium and < 0.04 mg of sodium.
Sodium Free
This product is free from artificial colours, flavours and preservatives, lactose, sodium and yeast.
Additional Information
KEEP AT ROOM TEMPERATURE IN A DARK DRY PLACE.
Helps relieve osteoarthritic symptoms and joint pain Helps to build healthy cartilage and protect against deterioration
{Caplet} Actual Size
400586.01-00
Purity Life(R)
Product Details
Research Evidence
Compare Similar Products