Iron Tablets 65 mg
This product looks safe
- No ingredients exceed tolerable upper intake levels
- Limited research evidence available for ingredients
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Suggested Use: Adults: Take one (1) tablet daily with or after a meal as a dietary supplement, or as recommended by your healthcare provider.
⚠️ Warnings & Precautions
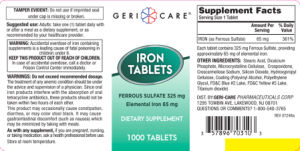
Tamper Evident: Do not use if imprinted seal under cap is missing or broken.
Warning: Accidental overdose of iron containing supplements is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or Poison Control Center immediately.
Warnings: Do not exceed recommended dosage. The treatment of any anemic condition should be under the advice and supervision of a physician.
Since oral iron products interfere with the absorption of oral tetracycline antibiotics, these products should not be taken within two hours of each other.
This product may occasionally cause constipation, diarrhea, or may color stool black. It may cause gastrointestinal discomfort (such as nausea) which may be minimized by taking with meals.
As with any supplement, if you are pregnant, nursing, or taking medication, ask a health professional before use.
🧪 Formulation Notes
Ferrous Sulfate 325 mg Elemental Iron 65 mg
Each tablet contains 325 mg Ferrous Sulfate, providing approximately 65 mg of elemental Iron.
Additional Information
Store at room temperature
Questions or comments? 1-800-540-3765
Product Details
Research Evidence
Compare Similar Products