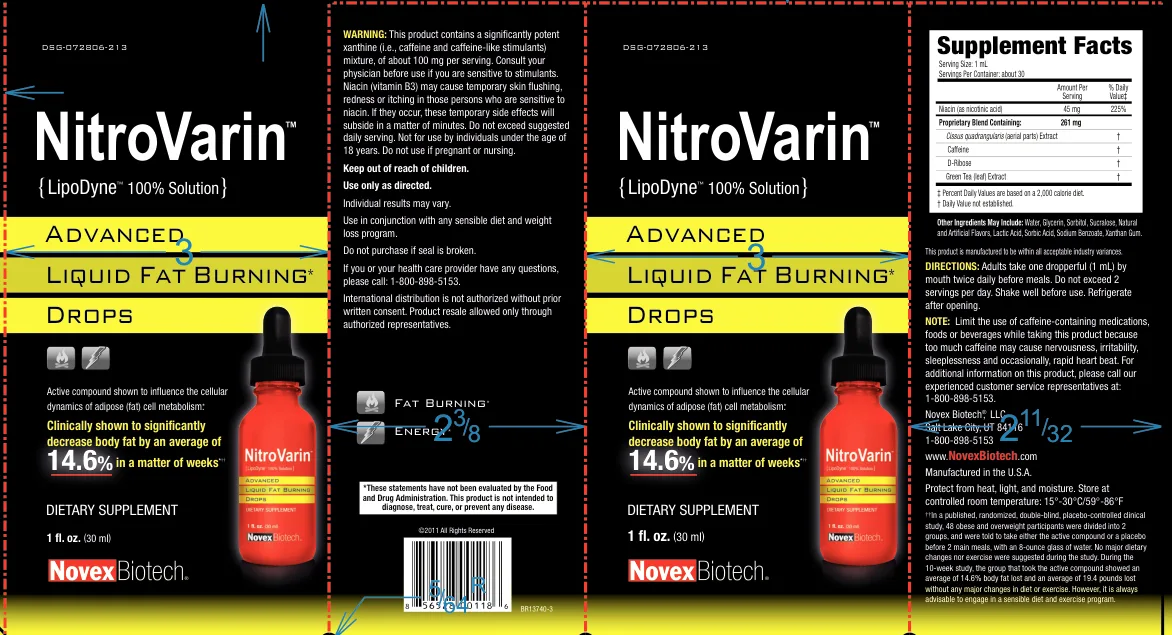
NitroVarin
Review before taking
- Niacin: 45mg is 1.3× the Tolerable Upper Intake Level (35mg)
- Contains proprietary blend — exact ingredient amounts not disclosed
- 50% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Safety Alerts
The product's niacin level exceeds the recommended maximum daily limit, and it uses a proprietary blend that does not disclose the exact amounts of all ingredients.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
DIRECTIONS: Adults take one dropperful (1 mL) by mouth twice daily before meals. Do not exceed 2 servings per day. Shake well before use. Refrigerate after opening.
⚠️ Warnings & Precautions
Keep out of reach of children.
Not for use by individuals under the age of 18 years. Do not use if pregnant or nursing.
Consult your physician before use if you are sensitive to stimulants.
Do not purchase if seal is broken.
Niacin (vitamin B3) may cause temporary skin flushing, redness or itching in those persons who are sensitive to niacin. If they occur, these temporary side effects will subside in a matter of minutes. Do not exceed suggested daily serving.
NOTE: Limit the use of caffeine-containing medications, foods or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness and occasionally, rapid heart beat. For additional information on this product, please call our experienced customer service representatives at: 1-800-898-5153.
WARNING: This product contains a significantly potent xanthine (i.e., caffeine and caffeine-like stimulants) mixture, of about 100 mg per serving.
Additional Information
Protect from heat, light, and moisture. Store at controlled room temperature: 15°-30°C/59°-86°F
††In a published, randomized, double-blind, placebo-controlled clinical study, 48 obese and overweight participants were divided into 2 groups, and were told to take either the active compound or a placebo before 2 main meals, with an 8-ounce glass of water. No major dietary changes nor exercise were suggested during the study. During the 10-week study, the group that took the active compound showed an average of 14.6% body fat lost and an average of 19.4 pounds lost without any major changes in diet or exercise. However, it is always advisable to engage in a sensible diet and exercise program.
©2011 All Rights Reserved
Active compound shown to influence the cellular dynamics of adipose (fat) cell metabolism.*
Advanced Liquid Fat Burning* Drops
BR13740-3
Clinically shown to significantly decrease body fat by an average of 14.6% in a matter of weeks*††
DSG-072806-213
Fat Burning* Energy*
If you or your health care provider have any questions, please call: 1-800-898-5153.
Individual results may vary.
International distribution is not authorized without prior written consent. Product resale allowed only through authorized representatives.
This product is manufactured to be within all acceptable industry variances.
Use in conjunction with any sensible diet and weight loss program.
Use only as directed.
Product Details
Research Evidence
Compare Similar Products