Calcium 600 mg
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 50% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
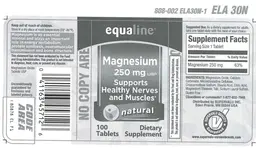
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Suggested use: Adults: Take one (1) tablet up to two times daily with food or as directed by your physician. Take with a full glass of water.
⚠️ Warnings & Precautions
Not formulated for children.
Do not exceed suggested use.
As with any supplement, if you are pregnant or nursing a baby, taking any medication, or have a medical condition, consult your doctor before using this product.
Keep out of reach of children.
Do not use if printed seal under cap is broken or missing
🧪 Formulation Notes
600 mg Calcium with 800 IU Vitamin D3
Additional Information
Compare to Caltrate 600+D3 ingredients Quality guaranteed
Store at room temperature. Keep bottle tightly closed.
This product is not manufactured or distributed by Pfizer, marketer of Caltrate 600+D3.
Our promise Quality & satisfaction 100% guaranteed or your money back.
Product Details
Research Evidence
Compare Similar Products