L2
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 50% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
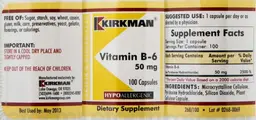
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Suggested use: As a dietary supplement for adults, take each serving of 4 capsules with 12-16 ounces of water. Use for 4-5 days at a time, then discontinue use for a minimum or 7 days before retaking the product. Please note: We highly recommend drinking a minimum of 6 glasses of water daily. Morning: Take 4 capsules with food. Mid-afternoon: Take 4 capsules with food.
⚠️ Warnings & Precautions
Warning: This product is only intended for healthy adults, 18 years of age or older.
Do not use if pregnant or nursing. Before using this product, consult a licensed qualified health care professional, including but not limited to, if: you are taking any other dietary supplement, prescription drug or over-the-counter medication such as for the treatment of diabetes or hyperglycemia; or if, you suspect you have or have been treated for, diagnosed with or have a family history of, any medical condition, including but not limited to: high or low blood pressure, diabetes, allergies to the Asteraceae/Compositae family, pancreatitis, anxiety, cardiovascular, psychiatric or seizure disorders, cardiac arrhythmia, stroke, heart, liver, kidney or thyroid disease, or difficulty urinating due to prostate enlargement.
Use only as directed. Exceeding recommendations for suggested use may cause adverse side effects, including but not limited to, weakness, muscle cramps, skin rash, diarrhea, dehydration, dizziness, or joint pain. If you experience these or any other adverse reaction to this product, immediately discontinue use and contact a medical doctor. Discontinue use 2 weeks prior to surgery. do not use if safety seal is broken or missing.
Keep out of reach of children
Additional Information
Store in a cool dry place.
101682
Water loss Shed excess water Tighten, tone, & define With mineral support
Manufactured in a GMP compliant facility
Product Details
Research Evidence
Compare Similar Products