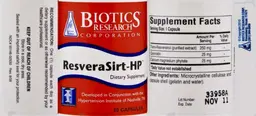
Trans-Resveratrol 250 Milligrams
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 100% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
1 capsule 1-2x daily Suggested Use: As a dietary supplement, take 1 capsule with your favorite beverage, 1-2 times daily or as directed by a physician.
⚠️ Warnings & Precautions
Warning: NOT INTENDED FOR USE BY PERSONS UNDER THE AGE OF 18. KEEP OUT OF THE REACH OF CHILDREN.
If you are pregnant, breast feeding, have known medical conditions (including kidney or liver disease) or are taking prescription or OTC medication(s) consult with your health care practitioner before using this product.
🧪 Formulation Notes
VEGETARIAN
~ ALLERGEN FREE ~ NON-GMO ~ GLUTEN FREE ~ BSE/TSE FREE ~ NON-IRR/ETO
ALLERGEN FREE
No fillers ~ No Excipients ~ No additives
Zero fillers or excipients inferior ingredients banned substances added sugars proprietary blends hidden ingredients
Free of Harmful Impurities
Kosher, Vegetarian, Hypoallergenic, Non-GMO, Gluten Free BSE/TSE Free and Non-GMO.
Kosher, Vegetarian, Hypoallergenic, Non-GMO, Gluten Free BSE/TSE Free and Non-GMO.
Kosher, Vegetarian, Hypoallergenic, Non-GMO, Gluten Free BSE/TSE Free and Non-GMO.
Kosher, Vegetarian, Hypoallergenic, Non-GMO, Gluten Free BSE/TSE Free and Non-GMO. No added sugars, salt, preservatives, salicylates, or artificial color, flavor or fragrance.
Additional Information
Manufactured in a GMP & FDA inspected facility
ANTIOXIDANT
ANTIOXIDANT & CARDIOVASCULAR SUPPORT
Pharmaceutical Grade HPLC Lab Tested for Potency
150 Servings 250 Milligrams
TRUE LABEL FULL LABEL DISCLOSURE
OU
Made in a GMP & FDA inspected facility
STORE IN A COOL DRY PLACE.
24461-006 B20079-002
Made in USA
Vegetable capsule
Since 1996
Quality & purity since 1996. Nutrabio manufactures in our own FDA registered & inspected. GMP facility complying with FDA 21 CFR Part 111 regulations.
Product Details
Research Evidence
Compare Similar Products