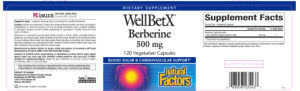
Berberine HCL Capsules 380 mg
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 100% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Suggested Use: As a dietary supplement, take 2 capsules twice daily, preferably 20 minutes before a meal.
⚠️ Warnings & Precautions
Disclaimer: Always consult with a qualified health physician before taking any new dietary supplement.
Warning: Keep out of reach of children. Do not take this or any other supplement if under the age of 18, pregnant or nursing a baby, or if you have any known or suspected medical conditions, and/or taking prescription drug(s) or OTC medication(s).
Do not take this or any other supplement if under the age of 18, pregnant or nursing a baby, or if you have any known or suspected medical conditions, and/or taking prescription drug(s) or OTC medication(s).
Additional Information
Storage: Store in a cool and dry place. Keep away from direct sunlight and heat.
Product Details
Research Evidence
Compare Similar Products