Glucosamine HCl
This product looks safe
- No ingredients exceed tolerable upper intake levels
- 33% of ingredients have research evidence
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
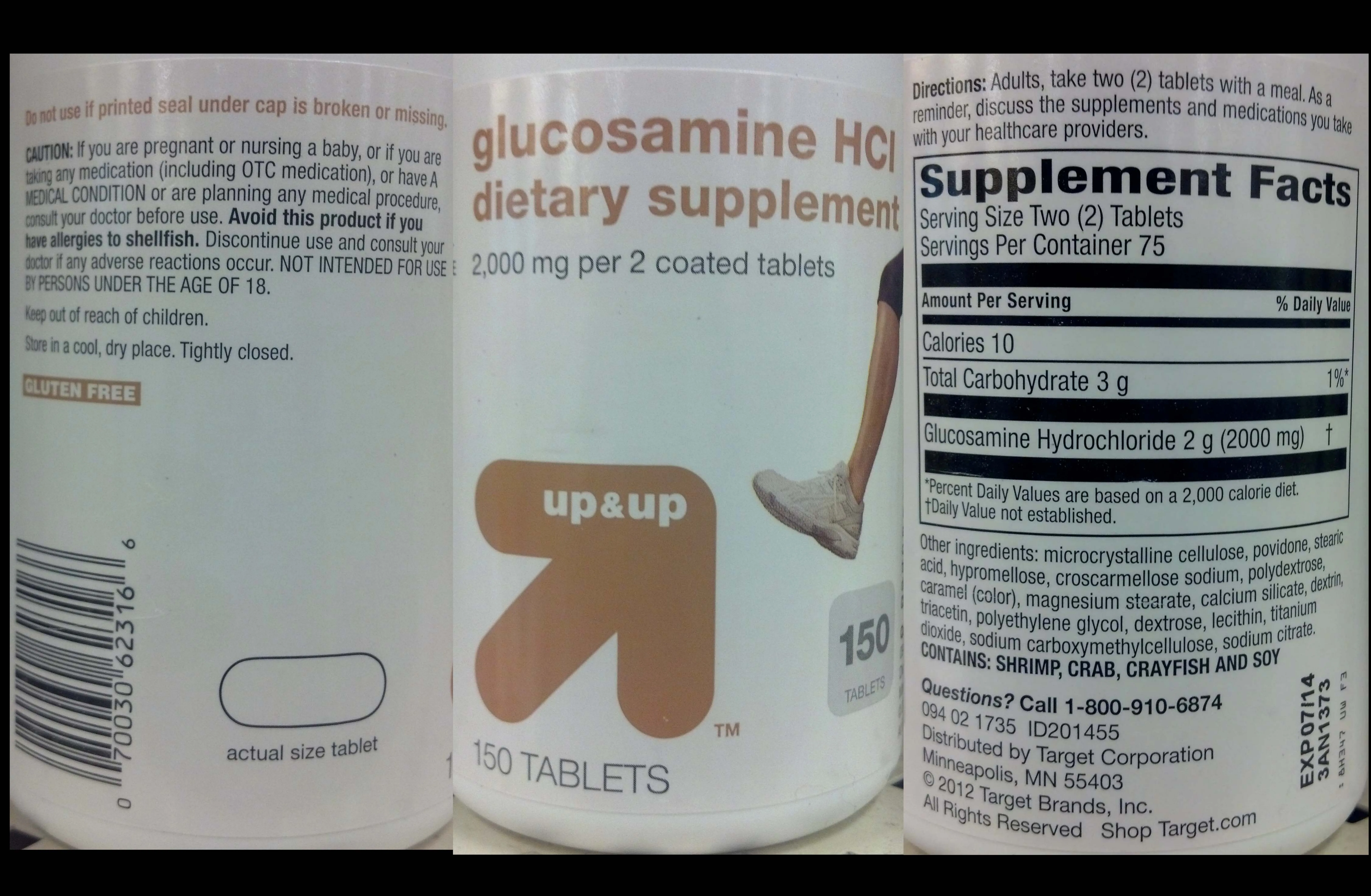
Directions: Adults, take two (2) tablets with a meal.
⚠️ Warnings & Precautions
Avoid this product if you have allergies to shellfish.
Discontinue use and consult your doctor if any adverse reactions occur.
NOT INTENDED FOR USE BY PERSONS UNDER THE AGE OF 18. Keep out of reach of children.
As a reminder, discuss the supplements and medications you take with your healthcare providers.
Do not use if printed seal under cap is broken or missing.
CAUTION: If you are pregnant or nursing a baby, or if you are taking any medication (including OTC medication), or have A MEDICAL CONDITION or are planning any medical procedure, consult your doctor before use.
🧪 Formulation Notes
GLUTEN FREE
CONTAINS: SHRIMP, CRAB, CRAYFISH AND SOY
Additional Information
actual size tablet
094 02 1735 : 8H347 UW F3
EXP07/14 3AN1373
Shop Target.com
Store in a cool, dry place. Tightly closed.
2,000 mg per 2 coated tablets
ID201455
Product Details
Research Evidence
Compare Similar Products