Artichoke Extract 500 mg
This product looks safe
- No ingredients exceed tolerable upper intake levels
- Limited research evidence available for ingredients
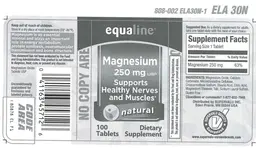
Product Label
Have a supplement label? Find it here.
Upload a photo of any supplement label to search our database of 92,312 products.
Label Data
Supplement Facts — Evidence Check
Other Ingredients
Label Claims — Verification
Target Groups
Product Information
📋 Directions for Use
Suggested use: 1 tablet daily with a meal.
⚠️ Warnings & Precautions
Note: if you are pregnant, may become pregnant, or breastfeeding, consult your health care professional before using this product.
Caution: do not take this product if you have gallstones or a bile duct obstruction, or if you are allergic to ragweed, chrysanthemums, marigolds or daisies.
Do not use if either tamper-evident seal is broken or missing.
Keep out of the reach of children.
🧪 Formulation Notes
For liver and digestive support
Artichoke extract is used throughout Europe to support liver and gallbladder function and relieve occasional indigestion. It works partly by increasing bile production, which may support increased fat metabolism. Planetary Herbals Artichoke extract is standardized to 5% cynarins for assured potency of this important compound.
Additional Information
Approved by Michael Tierra L.Ac, O.M.D Michael Tierra
Store in a cool, dry place.
Product Details
Research Evidence
Compare Similar Products